Acute Pancreatitis
2012 Revised Atlanta Classification of Acute Pancreatitis
Thomas Bollen, Marieke Hazewinkel and Robin Smithuis
Radiology department of the St. Antonius hospital, Nieuwegein, the Medical Centre Alkmaar and the Rijnland hospital, Leiderdorp, the Netherlands
Publicationdate
The 2012 Revised Atlanta Classification of acute pancreatitis enables standardized reporting which is helpful for communication between clinicians and for research.
A standardized template for reporting CT images is given to describe the local complications of acute pancreatitis.
Introduction
Temporally, two phases of acute pancreatitis are identified in the Revised Atlanta Classification:
- Early - first week
Only clinical parameters are important for treatment planning and are determined by the systemic inflammatory response syndrome - SIRS, which can lead to organ failure. - Late - after the first week
Morphologic criteria based on CT findings combined with clinical parameters determine the care of the patient.
The severity is classified into three categories based on clinical and morphologic findings:
- Mild - No organ failure and no local or systemic complications.
- Moderate - Presence of transient organ failure less than 48h and/or presence of local complications.
- Severe - Persistent organ failure > 48 hour.
15-20% of cases.
Morphologically, there are two types of acute pancreatitis:
- Acute oedematous or interstitial pancreatitis.
- Acute necrotizing pancreatitis.
- Usually the necrosis involves both the pancreas and the peripancreatic tissues.
- Less commonly only the peripancreatic tissues.
- Rarely only the pancreatic parenchyma.
Diagnosis of Acute Pancreatitis
The diagnosis of acute pancreatitis requires two of the following three features:
- Abdominal pain consistent with acute pancreatitis:
Acute onset of persistent, severe, epigastric pain often radiating to the back. - Serum lipase or amylase activity at least three times greater than the upper limit of normal.
- Characteristic findings of acute pancreatitis on contrast-enhanced CT (CECT) and less commonly MRI or US.
The diagnosis is usually established when there is a combination of abdominal pain and elevated pancreatic enzymes and CECT is not required, unless there is uncertainty about the diagnosis.
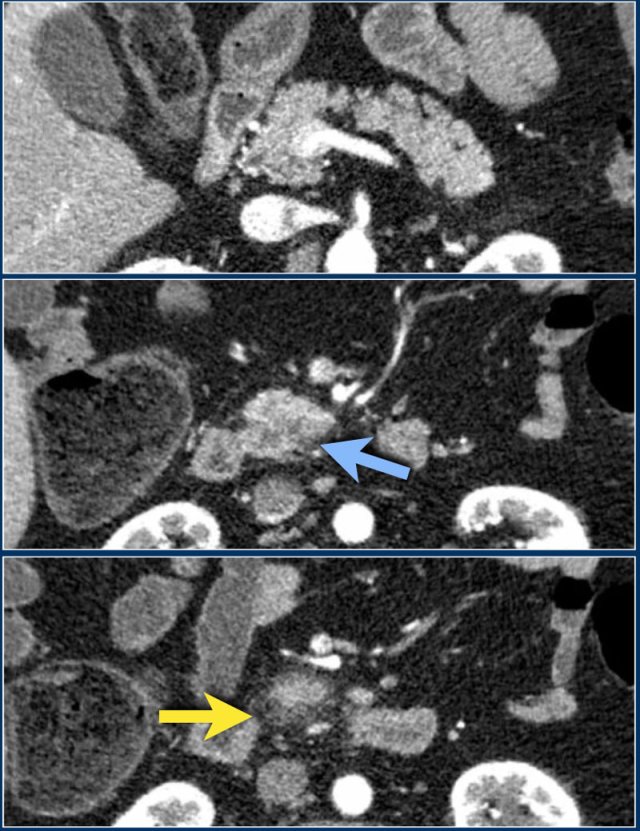
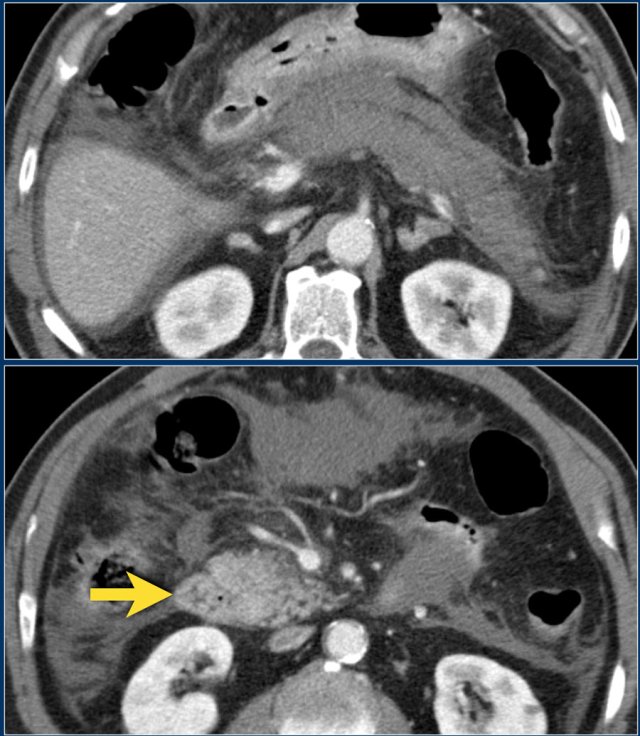
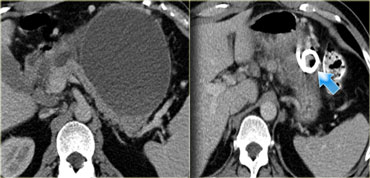
These images are of a patient who presented with acute severe epigastric pain very suggestive of acute pancreatitis.
However the amylase level was within normal levels.
A CECT was performed.
The findings are:
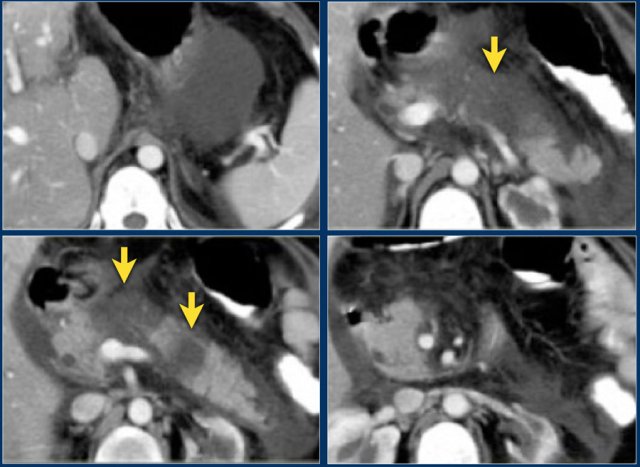
- Most of the pancreas is normal.
- Some edema of the uncinate process of the pancreatic head (blue arrow).
- Edema in the peripancreatic fat (yellow arrow), consistent with interstitial pancreatitis.
There were no complications and the diagnosis of mild pancreatitis was made.
Clinical outcome
Early severity stratification of acute pancreatitis is important to identify patients with the highest morbidity.
These patients may benefit from timely transfer to the intensive care unit or tertiary referral centre.
Mild pancreatitis
These patients have no organ failure.
Most of them have no fluid collections and no necrosis.
These patients usually recover by the end of the first week.
Moderate severe and severe pancreatitis
The clinical condition of the patient is determined by the host response to the pancreatitis.
Cytokine cascades result in a systemic inflammatory response syndrome (SIRS), which increases the risk of organ failure.
The presence of organ failure is determined by respiratory (pO2↓), renal (creatinine↑) and cardiovascular failure (blood pressure↓).
The extent of morphologic changes like necrosis and fluid collections is not directly proportional to the severity of organ failure.
Many of these patients however will have necrotizing pancreatitis and the mortality increases when the necrosis becomes infected.
Atlanta Classification of Fluid Collections
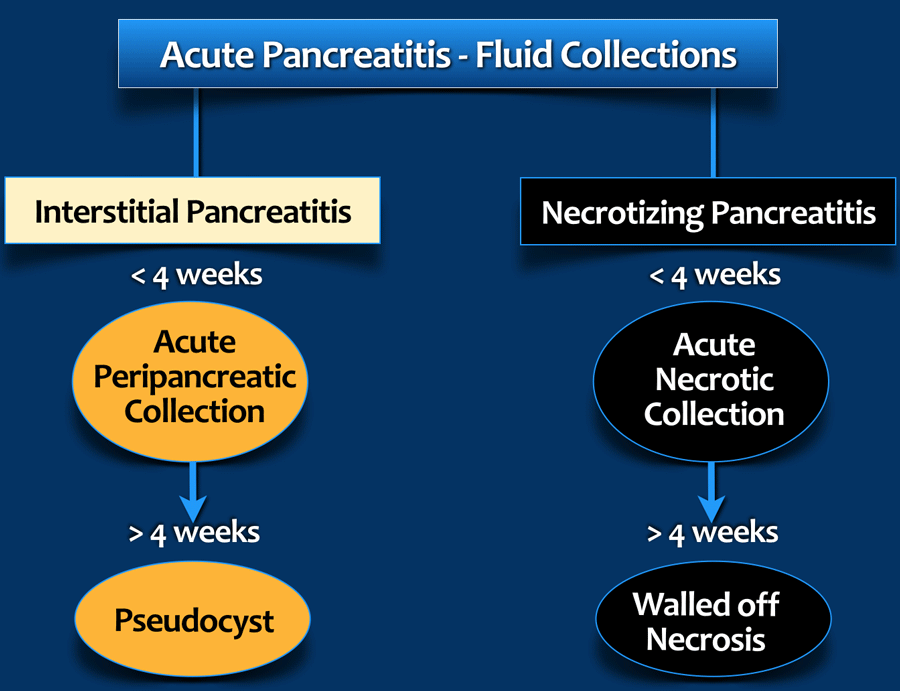
The 2012 Revised Atlanta Classification discerns 4 types of peripancreatic fluid collections in acute pancreatitis depending on the content, degree of encapsulation and time.
-
Content
- Fluid only in acute peripancreatic fluid collection (APFC) and Pseudocyst.
- Mixture of fluid and necrotic material in acute necrotic collection (ANC) and walled-off-necrosis (WON).
-
Degree of encapsulation
- None or partial wall in APFC and ANC.
- Complete encapsulation in pseudocyst and WON.
-
Time
- Within 4 weeks: APFC and ANC
- After 4 weeks: pseudocysts and WON. It takes about 4 weeks for a capsule to form.
On CT, the discrimination between an APFC and ANC may be difficult, especially in the first weeks and the term "indeterminate peripancreatic collections" can be used.
All these collections may remain sterile or become infected.
Infection is rare during the first week.
The table summarizes the CT criteria for pancreatic and peripancreatic fluid collections in acute pancreatitis.
- APFC
Acute Peripancreatic Fluid Collections contain fluid only and are not or only partially encapsulated. They are seen within 4 weeks in interstitial pancreatitis. Frequently they regress spontaneously. - ANC
Acute Necrotic Collections contain a mixture of fluid and necrotic material. They are not or only partially encapsulated. They are seen within 4 weeks in necrotizing pancreatitis. - Pseudocyst
After 4 weeks in interstitial pancreatitis. This fluid collection is encapsulated. Pseudocysts are uncommon in acute pancreatitis. Most persistent fluid collections also contain some necrotic material. - WON
After 4 weeks most necrotic collections are fully encapsulated and are called Walled-off Necrosis (WON).
CT severity index
The CT severity index (CTSI) combines the Balthazar grade (0-4 points) with the extent of pancreatic necrosis (0-6 points) on a 10-point severity scale.
On the day of admission, scoring systems based on imaging do not outperform scoring systems based on clinical and biochemical parameters with regard to predicting clinical outcome.
Therefore, performing CT on day of admission solely for prediction purposes is not recommended.
Imaging
CT
CT is the imaging modality of choice for the diagnosis and staging of acute pancreatitis and its complications.
Ultrasound and ERCP with sphincterotomy and stone extraction play an important role in biliary pancreatitis.
Since the diagnosis of acute pancreatitis is usually made on clinical and laboratory findings, an early CT is only recommended when the diagnosis is uncertain, or in case of suspected early complications such as bowel perforation or ischemia.
An early CT may be misleading regarding the morphologic severity of the pancreatitis, because it may underestimate the presence and amount of necrosis.
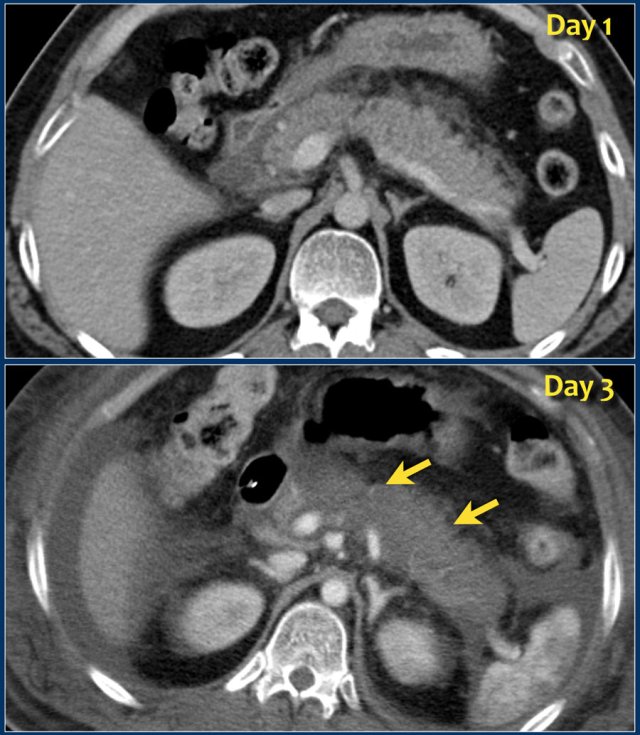
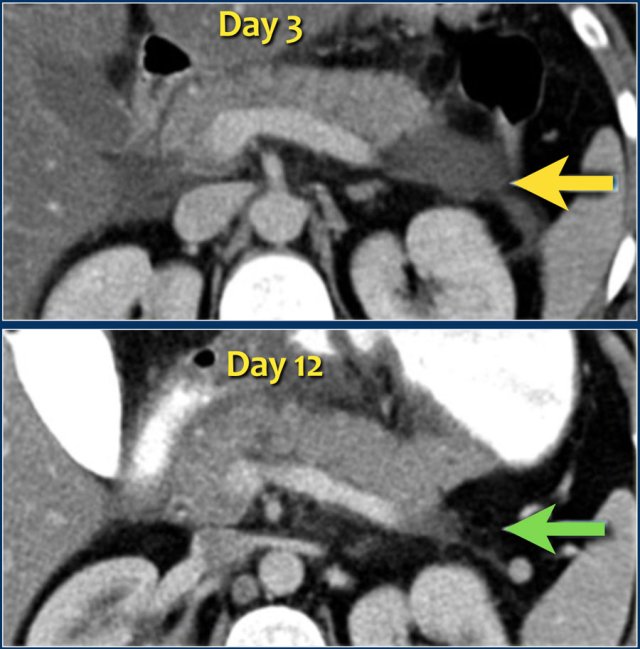
The images show a normally enhancing pancreas on day 1.
As the patient's condition worsened, a second CT was performed on day 3.
Notice how the greater part of the pancreatic body and tail no longer enhances indicating necrotizing pancreatitis (arrows).
The first CT underestimated the severity of the pancreatitis.
This patient died on day 5 due to severe SIRS and multiple organ failure.
Interstitial pancreatitis
Morphologically there are 2 types of acute pancreatitis - interstitial or oedematous pancreatitis and necrotizing pancreatitis..
Here an example of interstitial pancreatitis.
There is normal enhancement of the entire pancreatic gland with only mild surrounding fatty infiltration.
There are no fluid collections and there is no necrosis of the pancreatic parenchyma.
CTSI: 2 points.
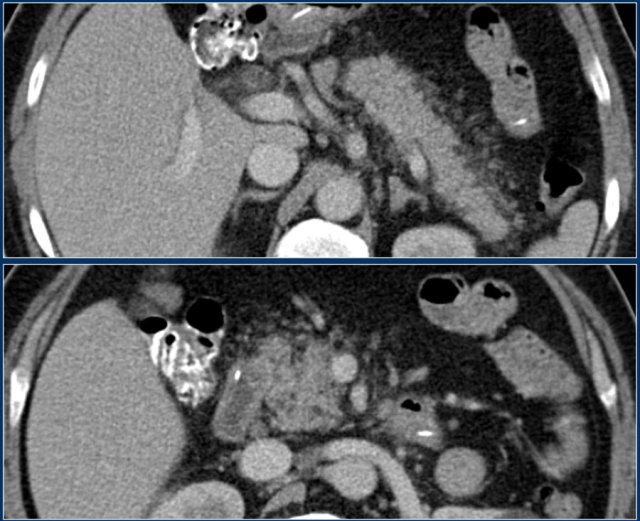
Here another case of interstitial pancreatitis without necrosis.
Scroll through the images.
The pancreas is swollen and there is peripancreatic inflammation (2 points).
No fluid collections.
No pancreatic necrosis.
CTSI: 2 points
Necrotizing Pancreatitis
Necrosis of pancreatic parenchyma or peripancreatic tissues occurs in 10-15 % of patients.
It is characterized by a protracted clinical course, a high incidence of local complications, and a high mortality rate.
There are 3 subtypes of necrotizing pancreatitis:
- Necrosis of both pancreatic parenchyma and peripancreatic tissues (most common).
- Necrosis of only extrapancreatic tissue without necrosis of pancreatic parenchyma (less common).
- Necrosis of pancreatic parenchyma without surrounding necrosis of peripancreatic tissue (very rare).
Necrosis of the pancreatic parenchyma can be diagnozed on a contrast-enhanced CT ⩾ 72 hours.
Necrosis of peripancreatic tissue can be vary difficult to diagnose, but is suspected when the collection is inhomogeneous, i.e. various densities on CT..
The CT shows an acute necrotizing pancreatitis.
The body and tail of the pancreas do not enhance.
There is normal enhancement of the pancreatic head (arrow).
More than 50% of the pancreas is necrotic and there are at least two collections.
CTSI: 4 + 6 = 10 points.
MRI
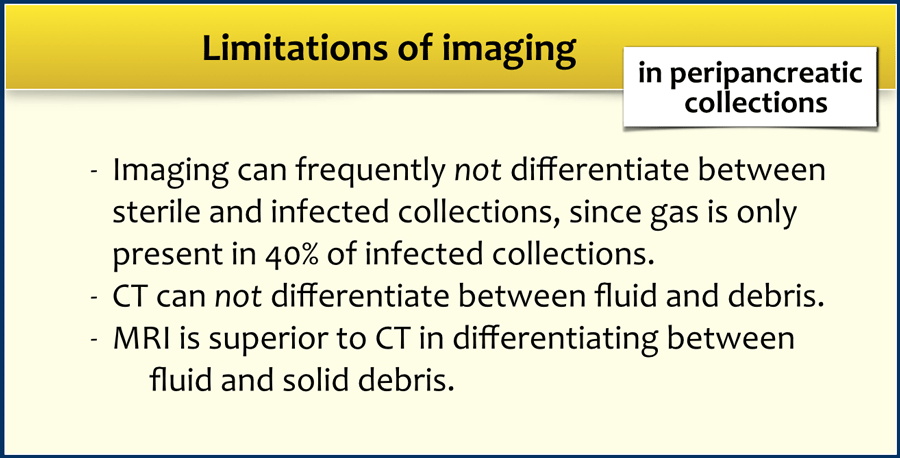
MRI is superior to CT in differentiating between fluid and solid necrotic debris.
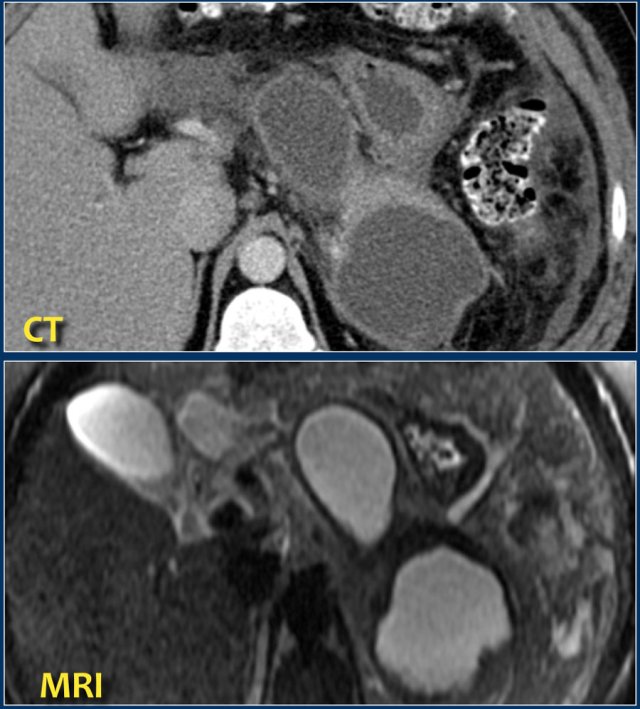
Here a patient with several homogeneous peripancreatic collections on CT.
These collections also show homogeneous high signal intensity on a fat-suppressed T2-weighted MRI image, are fully encapsulated and contain clear fluid (i.e. pseudocysts).
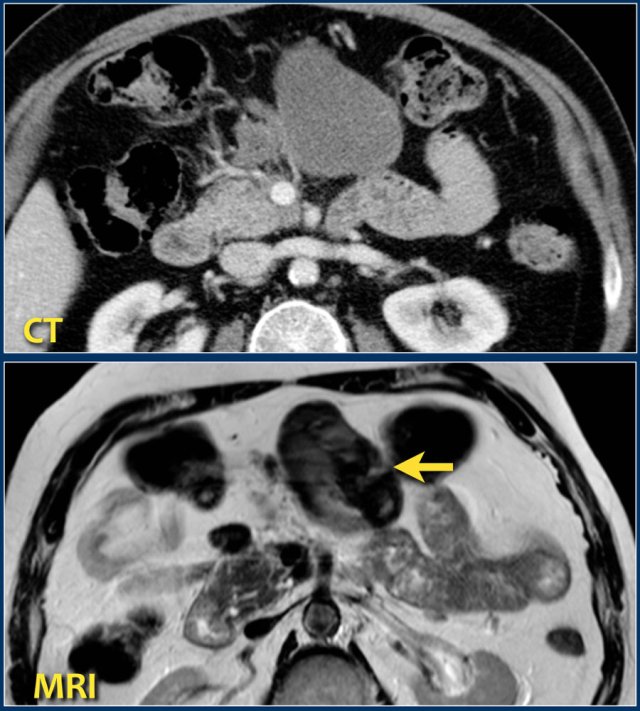
This patient had an acute necrotizing pancreatitis with onset 2 months earlier.
The CT-image shows a homogeneous peripancreatic collection in the transverse mesocolon (arrow).
A T2-weighted MRI sequence shows that the collection has a low signal intensity (arrow).
Most likely this is necrotic fat tissue (i.e. sterile necrosis or walled-off necrosis).
This patient had no fever or signs of sepsis.
Endoscopic or percutaneous drainage would have little or no effect on its size, but increases the risk of infection.
Peripancreatic Collections
Acute Peripancreatic Fluid Collection - APFC
Intraabdominal fluid collections and collections of necrotic tissue are common in acute pancreatitis.
These collections develop early in the course of acute pancreatitis.
In the early stage, such a collection does not have a wall or capsule.
Preferred locations of fluid collections are:
- Lesser sac
- Anterior and posterior pararenal space of the retroperitoneum.
- Transverse mesocolon
- Small bowel mesentery.
These collections are the result of the release of activated pancreatic enzymes which also cause necrosis of the surrounding tissues.
This explains why many of these collections harbor solid necrotic debris.
About 50% of these collections show spontaneous regression (figure).
The remaining 50% either remain stable or increase and undergo organization and demarcation with liquefaction.
They may remain sterile or develop infection.
The images show spontaneous regression of an acute peripancreatic fluid collection (APFC).
Acute Necrotic Collection - ANC
Study the image.
What are the findings?
Then continue reading.
The findings are:
- Necrosis of the pancreas
- Inhomogeneous collection in the peripancreatic tissue
- No wall
We can conclude that this is an acute necrotic collection - ANC.
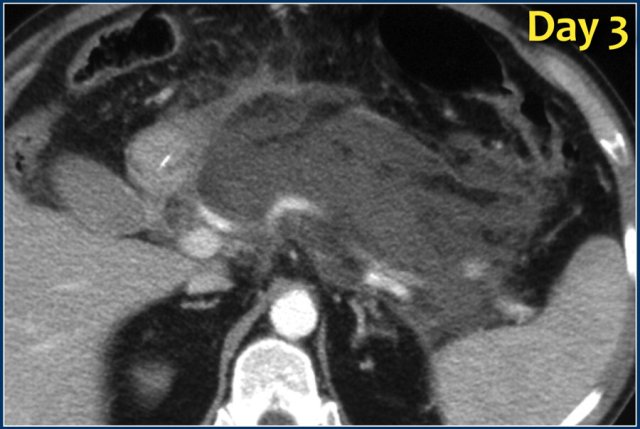
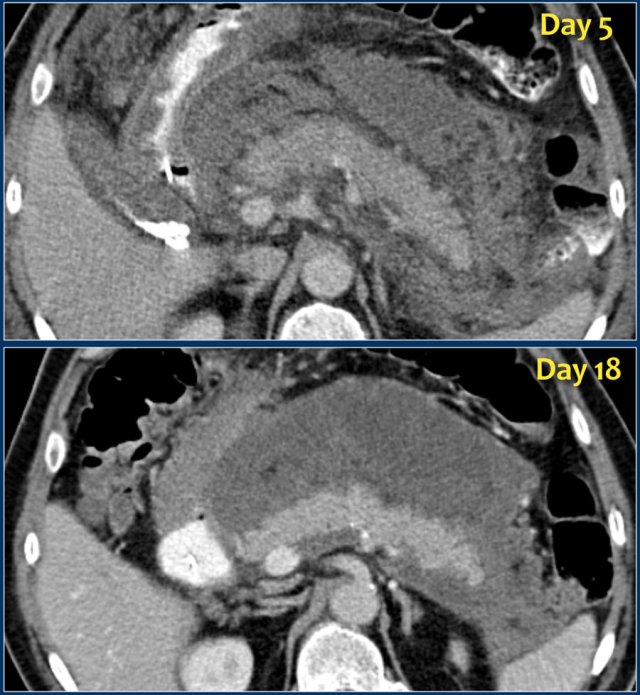
ANC (2)
Study the images and then continue reading.
What are the findings?
The findings are:
- Normal enhancement of the entire pancreas.
- Extensive peripancreatic collections, which have liquid and non-liquid densities on CT.
- There are at least two collections, but no pancreatic parenchymal necrosis (CTSI: 4).
- On day 18 there is expansion of the peripancreatic collections and an incomplete wall is present.
On day 5 this collection can be diagnosed as probable acute necrotic collection.
On day 18 an incomplete wall is present, but we can assume that in a couple of days this will be a walled-of-necrosis with a complete wall.
When peripancreatic collections persist or increase, it is usually due to the presence of fat necrosis.
Because fat does not enhance on CT, the diagnosis of fat necrosis can be difficult.
Necrosis can be diagnosed with MRI, which of course should only be performed if it has direct clinical implications.
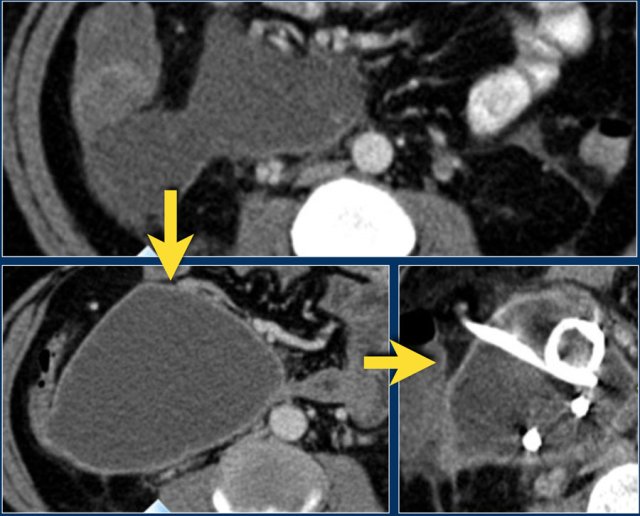
Pseudocyst
This patient presented with a gastric outlet obstruction 2 months after an episode of acute pancreatitis.
There is a homogeneous well-demarcated peripancreatic collection in the lesser sac, which abuts the stomach and the pancreas.
The patient did not have fever.
The collection underwent successful percutaneous drainage, which showed clear fluid with high amylase and subsequently resolved along with the patient's symptoms.
Therefore, this collection proved to be a true pancreatic pseudocyst.
A Pseudocyst is a collection of pancreatic juice or fluid enclosed by a complete wall of fibrous tissue
It occurs in interstitial pancreatitis and the absence of necrotic tissue is imperative for its diagnosis.
Communication with the pancreatic duct may be present.
A pseudocyst requires 4 or more weeks to develop.
The differential diagnosis includes walled-off necrosis and sometimes a pseudoaneurysm or even a cystic tumor.
Most often, they occur in the lesser sac.
Most collections that persist after 4 weeks are walled-of-necrosis.
True pseudocysts are uncommon, since most acute peripancreatic fluid collections resolve within 4 weeks.
Walled-off Necrosis - WON
Based on CT alone it is sometimes impossible to determine whether a collection contains fluid only or a mixture of fluid and necrotic tissue.
Consequently it is sometimes better to describe these as 'indeterminate peripancreatic collections'.
The images are of a patient with acute pancreatitis.
On the upper image is a collection in the area of the pancreatic head in the right anterior pararenal space.
At this stage, it is not possible to distinguish between an acute peripancreatic fluid collection and acute necrotic collection.
On a follow-up scan the collection in the right anterior pararenal space increased in size.
It has fluid density and a thin enhancing wall.
This can be a pseudocyst or walled-off-necrosis and it may or may not be infected.
The patient became septic and a percutaneous drainage was performed.
After drainage the collection barely diminished in size.
The patient underwent surgery and the collection was found to consist of necrotic debris, which was not appreciated on CT, hence this was a walled-off-necrosis and not a pseudocyst.
The necrotic debris was too viscous for successful percutaneous drainage.
Walled-off-necrosis (2)
These CT-images are of a patient on day 40.
This patient had central gland necrosis and now developed fever.
The CT shows a similar collection of fluid density to that of the patient with the pseudocyst, except for its pancreatic location.
The collection is homogeneous and well-demarcated with a thin wall abutting the stomach.
During endoscopic debridement this collection contained fluid and necrotic tissue, which was removed from the area of the pancreas.
Although the imaging characteristics in this case are similar to the patient with the pseudocyst, this proved to be infected walled-off-necrosis.
Walled-off-necrosis (3)
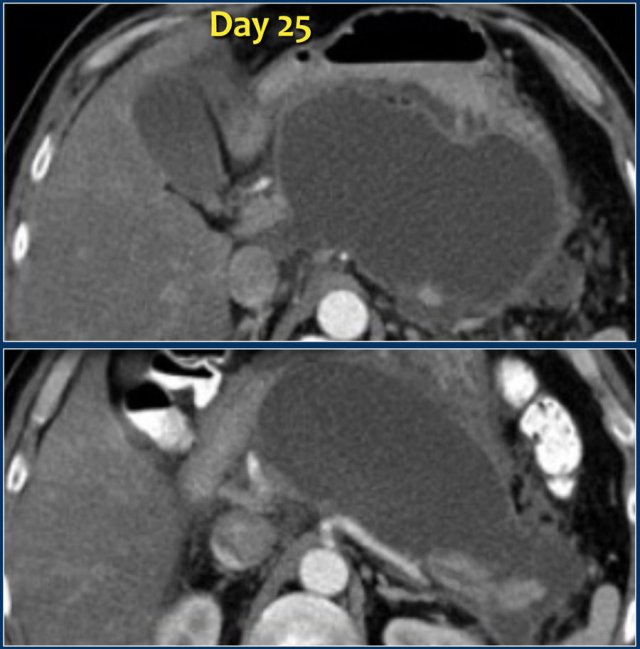
Here we see a homogeneous pancreatic and peripancreatic collection, well demarcated with an enhancing wall, on day 25 of an episode of acute necrotizing pancreatitis.
This patient had fever and multiple organ failure.
Therefore, this collection was suspected to be infected WON and not a pseudocyst.
At surgery, the collection contained much necrotic debris, which was not depicted on CT.
These cases illustrate that at times CT cannot reliably differentiate between collections that consist of fluid only and those that contain fluid and solid necrotic debris with or without infection.
Infected necrosis
Infected necrosis is:
- Infection of necrotic pancreatic parenchyma or extrapancreatic fatty tissue - i.e. infected ANC or infected WON, depending on degree of encapsulation.
- Usually occurs in the 2nd-4th week and rarely in the first week.
- Most severe local complication of acute necrotizing pancreatitis.
- Most common cause of death in patients with acute pancreatitis.
- Diagnose infected necrosis when there are gas bubbles on CT (seen in 40%) or when FNA is positive for bacteria.
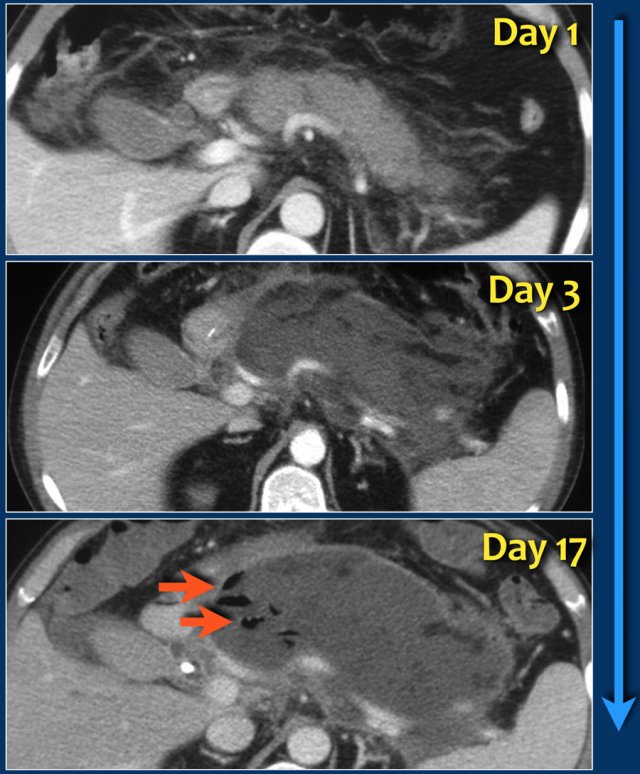
This case is a typical example of infected pancreatic necrosis.
- On day 1 there is enhancement of the pancreas and it just looks like a mild interstitial pancreatitis.
- On day 3 there is no enhancement of the pancreas, consistent with necrosis.
The necrosis also involves the peripancreatic tissue.
So this is an ANC - acute necrotic collection. - On day 17 there are gas bubbles in the necrotic collection consistent with infected pancreatic and peripancreatic necrosis.
A wall surrounds the collection.
The term pancreatic abcess is no longer used, since a collection of pus without necrotic tissue is extremely uncommon in acute pancreatitis.
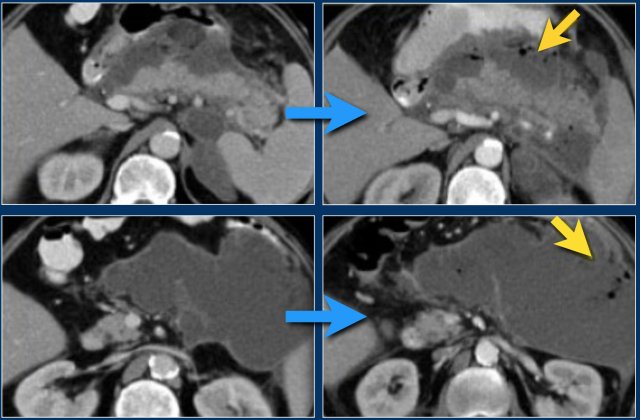
Infected necrosis (2)
In this patient there is normal enhancement of the pancreas with surrounding septated heterogeneous acute necrotic collections with fluid- and fat densities.
This is probably necrosis of the peripancreatic tissues.
Two weeks later there are gas bubbles in the peripancreatic collection consistent with an infected acute necrotic collection.
This patient underwent surgery.
The surgeon removed a large amount of necrotic tissue and estimated he had removed over 90% of the pancreas.
Continue with the next image.
Remarkably, a CT performed 6 months after surgery showed a normal pancreas.
This indicates that during surgery the differentiation between pancreatic necrosis and necrosis of the peripancreatic tissues is sometimes impossible.
The limitations of imaging are summarized in the table.
Central gland necrosis
Central gland necrosis is a specific form of necrotizing pancreatitis, representing full thickness necrosis between the pancreatic head and tail and is nearly always associated with disruption of the pancreatic duct.
This leads to persistent collections as the viable pancreatic tail continues to secrete pancreatic juices.
These collections mayreact poorly to endoscopic or percutaneous drainage.
Definitive treatment may require distal pancreatectomy or long-term endoscopic drainage.
Reporting - PANCODE
The Pancode system is a checklist for the description of acute pancreatitis and its complications.
Intervention
The current management of acute pancreatitis is to be conservative for as long as possible.
During the first two weeks patients with severe acute pancreatitis and multi-organ failure should be stabilized in the ICU.
Interventions should be delayed for as long as possible.
Many collections will remain sterile or resorb spontaneously.
Allow for demarcation of collections, which takes about 4 weeks.
FNA and Drainage
Once the clinical condition of the patient deteriorates and the patient is febrile, fine needle aspiration (FNA) can be used to differentiate between sterile and infected collections.
Important remarks concerning FNA:
- No role for FNA in early collections.
- Not transgastric route for diagnosis only.
- Be sure it is not a pseudoaneurysm
- Think ahead - What is the plan:
- Pus > tube placement.
- Indeterminate or clear > Gram stain, culture.
- No fluid > probably solid necrosis > install saline > culture.
Important remarks concerning Drainage:
- Indications for intervention of evolving peripancreatic collections should be based on full evaluation of clinical, lab, and imaging
- No role for drainage in early collections
- Can be used as a guide for surgical approach
Peripancreatic collections can be approached through the transhepatic (red arrow), transgastric (green arrow) or transabdominal (blue arrows) route, but the preferred approach is to stay in the retroperitoneal compartment (yellow arrows).
The retroperitoneal approach has some advantages:
- Same compartment as the pancreas.
- No contamination with intestinal flora.
- Gravity.
- Drain runs parallel to pancreatic bed.
- Route can be used to guide minimally invasive surgery.
Surgical intervention
In 2013, the IAP/APA evidence-based guideline for the management for acute pancreatitis was published.
The following recommendations were made:
- Indications for intervention in necrotizing pancreatitis are:
- Clinical suspicion or documented infected necrotizing pancreatitis with clinical deterioration
- Ongoing organ failure for several weeks after disease onset in the absence of documented infected necrotizing pancreatitis.
- Indications for intervention in sterile necrotizing pancreatitis are:
- Ongoing gastric outlet, intestinal, or biliary obstruction due to mass effect of walled-off necrosis (i.e. arbitrarily >4-8 weeks after onset of acute pancreatitis)
- Persistent debilitating symptoms in patients with walled-off necrosis without signs of infection (i.e. arbitrarily >8 weeks after onset of acute pancreatitis)
- Disconnected duct syndrome with persisting symptoms (e.g. pain, obstruction) collection(s) with walled-off necrosis.
- For patients with proven or suspected infected necrotizing pancreatitis, invasive intervention should be preferably delayed until at least 4 weeks after initial presentation to allow collections to become 'walled-off'.
- The optimal interventional strategy for patients with suspected or confirmed infected necrotizing pancreatitis is initial image-guided percutaneous (retroperitoneal) catheter drainage or endoscopic transluminal drainage, followed, if necessary, by endoscopic or surgical necrosectomy.
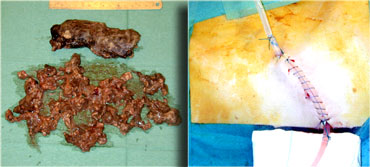 After removal of the necrotic tissue through videoscopic assisted retroperitoneal debridement, catheters are left in place
After removal of the necrotic tissue through videoscopic assisted retroperitoneal debridement, catheters are left in place
Take home messages
- Be familiar with the terminology in acute pancreatitis using the 2012 Revised Atlanta Classification.
- Morphologic severity of acute pancreatitis (including pancreatic parenchymal necrosis) can only be reliably assessed by imaging 72 hours after onset of symptoms.
- CT can not reliably differentiate between collections that consist of fluid only and those that contain solid necrotic debris.
In these cases MRI can be of additional value. - Avoid early drainage of collections and avoid introducing infection.